Date Published: August 24, 2022
The persistence of the global COVID-19 pandemic caused by the SARS-CoV-2 virus has continued to emphasize the need for point-of-care (POC) diagnostic tests for viral diagnosis. The most widely used tests, lateral flow assays used in rapid antigen tests, and reverse-transcriptase real-time polymerase chain reaction (RT-PCR), have been instrumental in mitigating the impact of new waves of the pandemic, but fail to provide both sensitive and rapid readout to patients. Here, we present a portable lens-free imaging system coupled with a particle agglutination assay as a novel biosensor for SARS-CoV-2. This sensor images and quantifies individual microbeads undergoing agglutination through a combination of computational imaging and deep learning as a way to detect levels of SARS-CoV-2 in a complex sample. SARS-CoV-2 pseudovirus in solution is incubated with acetyl cholinesterase 2 (ACE2)-functionalized microbeads then loaded into an inexpensive imaging chip. The sample is imaged in a portable in-line lens-free holographic microscope and an image is reconstructed from a pixel superresolved hologram. Images are analyzed by a deep-learning algorithm that distinguishes microbead agglutination from cell debris and viral particle aggregates, and agglutination is quantified based on the network output. We propose an assay procedure using two images which results in the accurate determination of viral concentrations greater than the limit of detection (LOD) of 1.27 × 103 copies per mL, with a tested dynamic range of 3 orders of magnitude, without yet reaching the upper limit. This biosensor can be used for fast SARS-CoV-2 diagnosis in low-resource POC settings and has the potential to mitigate the spread of future waves of the pandemic.
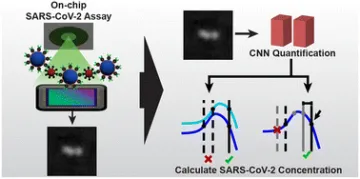
Diagram representing assay and quantification process to calculate SARS-CoV-2 concentration.
